- 1.
Siegel, R. L., Miller, K. D. & Jemal, A. Cancer statistics, 2019. CA Cancer J. Clin. 69, 7–34 (2019).
Article Google Scholar
- 2.
Fizazi K., et al. LATITUDE: A phase III, double-blind, randomized trial of androgen deprivation therapy with abiraterone acetate plus prednisone or placebos in newly diagnosed high-risk metastatic hormone-naive prostate cancer. J Clin Oncol 35, LBA3-LBA3 (2017).
- 3.
Zhang J., Cunningham J. J., Brown J. S., Gatenby R. A. Integrating evolutionary dynamics into treatment of metastatic castrate-resistant prostate cancer. Nat. Commun. 8, 1816 (2017).
- 4.
Wargo, A. R., Huijben, S., de Roode, J. C., Shepherd, J. & Read, A. F. Competitive release and facilitation of drug-resistant parasites after therapeutic chemotherapy in a rodent malaria model. Proc. Natl. Acad. Sci. USA 104, 19914–19919 (2007).
ADS CAS Article Google Scholar
- 5.
Crook, J. M. et al. Intermittent androgen suppression for rising PSA level after radiotherapy. N. Engl. J. Med 367, 895–903 (2012).
ADS CAS Article Google Scholar
- 6.
Baez, J. & Kuang, Y. Mathematical models of androgen resistance in prostate cancer patients under intermittent androgen suppression therapy. Appl. Sci. 6, 352 (2016).
Article Google Scholar
- 7.
Cunningham J. J., Brown J. S., Gatenby R. A., Stankova K. Optimal control to develop therapeutic strategies for metastatic castrate resistant prostate cancer. J. Theor. Biol. 459, 67–78 (2018).
- 8.
Hirata Y., et al. Intermittent androgen suppression: estimating parameters for individual patients based on initial PSA data in response to androgen deprivation therapy. PLoS ONE 10, e0130372 (2015).
- 9.
Hirata Y., Morino K., Akakura K., Higano C. S., Aihara K. Personalizing androgen suppression for prostate cancer using mathematical modeling. Sci. Rep. 8, 2673 (2018).
- 10.
Hirata, Y., Bruchovsky, N. & Aihara, K. Development of a mathematical model that predicts the outcome of hormone therapy for prostate cancer. J. Theor. Biol. 264, 517–527 (2010).
MathSciNet CAS Article Google Scholar
- 11.
Ideta, A. M., Tanaka, G., Takeuchi, T. & Aihara, Z. A mathematical model of intermittent androgen suppression for prostate cancer. J. Nonlinear Sci. 18, 593–614 (2008).
ADS CAS Article Google Scholar
- 12.
Morken, J. D., Packer, A., Everett, R. A., Nagy, J. D. & Kuang, Y. Mechanisms of resistance to intermittent androgen deprivation in patients with prostate cancer identified by a novel computational method. Cancer Res. 74, 3673–3683 (2014).
CAS Article Google Scholar
- 13.
Portz T., Kuang, Y., Nagy, J. A clinical data validated mathematical model of prostate cancer growth under intermittent androgen suppression therapy AIP Advances 2, 0110022 (2012).
- 14.
Elishmereni, M. et al. Predicting time to castration resistance in hormone sensitive prostate cancer by a personalization algorithm based on a mechanistic model integrating patient data. Prostate 76, 48–57 (2016).
CAS Article Google Scholar
- 15.
Collins, A. T. & Maitland, N. J. Prostate cancer stem cells. Eur. J. Cancer 42, 1213–1218 (2006).
CAS Article Google Scholar
- 16.
Enderling, H. Cancer stem cells: small subpopulation or evolving fraction? Integr. Biol. 7, 14–23 (2015).
Article Google Scholar
- 17.
Maitland, N. J. & Collins, A. T. Prostate cancer stem cells: a new target for therapy. J. Clin. Oncol. 26, 2862–2870 (2008).
Article Google Scholar
- 18.
Isaacs, J. T. & Coffey, D. S. Etiology and disease process of benign prostatic hyperplasia. Prostate Suppl. 2, 33–50 (1989).
CAS Article Google Scholar
- 19.
Collins, A. T., Berry, P. A., Hyde, C., Stower, M. J. & Maitland, N. J. Prospective identification of tumorigenic prostate cancer stem cells. Cancer Res 65, 10946–10951 (2005).
CAS Article Google Scholar
- 20.
Bruchovsky, N., Rennie, P. S., Coldman, A. J., Goldenberg, S. L., To M & Lawson, D. Effects of androgen withdrawal on the stem cell composition of the Shionogi carcinoma. Cancer Res. 50, 2275–2282 (1990).
CAS PubMed Google Scholar
- 21.
Lee, S. O. et al. New therapy targeting differential androgen receptor signaling in prostate cancer stem/progenitor vs. non-stem/progenitor cells. J. Mol. Cell Biol. 5, 14–26 (2013).
CAS Article Google Scholar
- 22.
Bruchovsky, N. et al. Final results of the Canadian prospective phase II trial of intermittent androgen suppression for men in biochemical recurrence after radiotherapy for locally advanced prostate cancer: clinical parameters. Cancer 107, 389–395 (2006).
Article Google Scholar
- 23.
Baxter W. C., Stanley J. S., Zhang Q., Smith W. D. Developing artificial neural network models of water treatment processes: a guide for utilities. J. Environ. Eng. Sci. 1, 201–211 (2002).
- 24.
Johnson, C. H. Circadian clocks and cell division: what’s the pacemaker? Cell Cycle 9, 3864–3873 (2010).
CAS Article Google Scholar
- 25.
Fawcett T. An introduction to ROC analysis. Pattern Recognit. Lett. 27, 861–874 (2005).
- 26.
Hussain, M. et al. Intermittent versus continuous androgen deprivation in prostate cancer. N. Engl. J. Med. 368, 1314–1325 (2013).
CAS Article Google Scholar
- 27.
Chi, K. N. et al. Apalutamide for metastatic, castration-sensitive prostate cancer. N. Engl. J. Med. 381, 13–24 (2019).
CAS Article Google Scholar
- 28.
Hoyle, A. P. et al. Abiraterone in “high-“ and “low-risk” metastatic hormone-sensitive prostate cancer. Eur. Urol. 76, 719–728 (2019).
CAS Article Google Scholar
- 29.
James, N. D. et al. Addition of docetaxel, zoledronic acid, or both to first-line long-term hormone therapy in prostate cancer (STAMPEDE): survival results from an adaptive, multiarm, multistage, platform randomised controlled trial. Lancet 387, 1163–1177 (2016).
CAS Article Google Scholar
- 30.
Kyriakopoulos, C. E. et al. Chemohormonal therapy in metastatic hormone-sensitive prostate cancer: long-term survival analysis of the randomized phase III E3805 CHAARTED trial. J. Clin. Oncol. 36, 1080–1087 (2018).
CAS Article Google Scholar
- 31.
Vale, C. L. et al. Addition of docetaxel or bisphosphonates to standard of care in men with localised or metastatic, hormone-sensitive prostate cancer: a systematic review and meta-analyses of aggregate data. Lancet Oncol. 17, 243–256 (2016).
CAS Article Google Scholar
- 32.
Singh, S. K. et al. Identification of a cancer stem cell in human brain tumors. Cancer Res. 63, 5821–5828 (2003).
CAS PubMed Google Scholar
- 33.
Boerrigter E., Groen L. N., Van Erp N. P., Verhaegh G. W., Schalken J. A. Clinical utility of emerging biomarkers in prostate cancer liquid biopsies. Expert. Rev. Mol. Diagn. 20, 219–230 (2020).
- 34.
Stankova, K., Brown, J. S., Dalton, W. S. & Gatenby, R. A. Optimizing cancer treatment using game theory: a review. JAMA Oncol. 5, 96–103 (2019).
Article Google Scholar
- 35.
Gravis, G. et al. Androgen deprivation therapy (ADT) plus docetaxel versus ADT alone in metastatic non castrate prostate cancer: impact of metastatic burden and long-term survival analysis of the randomized phase 3 GETUG-AFU15 trial. Eur. Urol. 70, 256–262 (2016).
CAS Article Google Scholar
- 36.
Brady R., Enderling H. Mathematical models of cancer: when to predict novel therapies, and when not to. Bull. Math Biol. 81, 3722–3731 (2019).
- 37.
Lee, D. K. et al. Progression of prostate cancer despite an extremely low serum level of prostate-specific antigen. Korean J. Urol. 51, 358–361 (2010).
Article Google Scholar
- 38.
Thompson, I. M. et al. Prevalence of prostate cancer among men with a prostate-specific antigen level < or =4.0 ng per milliliter. N. Engl. J. Med. 350, 2239–2246 (2004).
CAS Article Google Scholar
- 39.
Danila, D. C. et al. Circulating tumor cell number and prognosis in progressive castration-resistant prostate cancer. Clin. Cancer Res 13, 7053–7058 (2007).
CAS Article Google Scholar
- 40.
Aragon-Ching, J. B. et al. Circulating tumor cells in biochemical recurrence of prostate cancer. Clin. Genitourin. Cancer 13, e341–e345 (2015).
Article Google Scholar
- 41.
Tu, S. M. & Lin, S. H. Prostate cancer stem cells. Clin. Genitourin. Cancer 10, 69–76 (2012).
Article Google Scholar
- 42.
Glass, L. Synchronization and rhythmic processes in physiology. Nature 410, 277–284 (2001).
ADS CAS Article Google Scholar
- 43.
Matsu-Ura, T. et al. Intercellular coupling of the cell cycle and circadian clock in adult stem cell culture. Mol. Cell 64, 900–912 (2016).
CAS Article Google Scholar
- 44.
Banks H. T., Tran H. T. Mathematical and Experimental Modeling of Physical and Biological Processes. Chapman and Hall (2009).
- 45.
Olufsen, M. S. & Ottesen, J. T. A practical approach to parameter estimation applied to model predicting heart rate regulation. J. Math. Biol. 67, 39–68 (2013).
MathSciNet Article Google Scholar
- 46.
Poli, R., Kennedy, J. & Blackwell, T. Particle swarm optimization. Swarm Intell. 1, 33 (2007).
Article Google Scholar
- 47.
Jaworska D., Szliszka E. Targeting apoptotic activity against prostate cancer stem cells. Int. J. Mol. Sci. 18, pii: E1648 (2017).



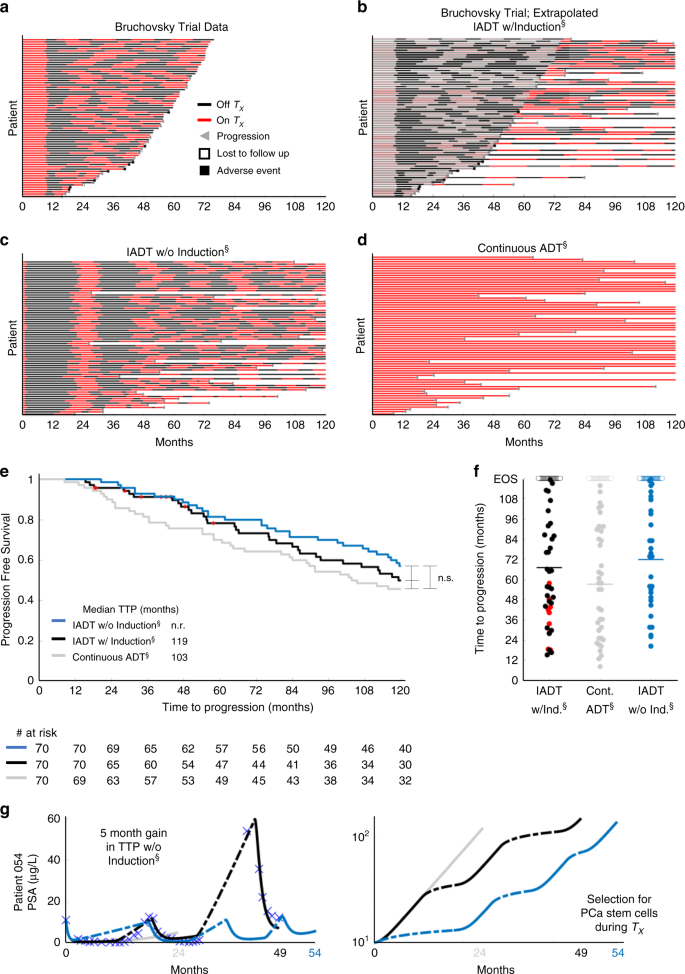
Plaats een reactie ...
1 Reactie op "Intermitterende hormoontherapie (IADT) geeft zelfde resultaten op progressievrije tijd en overall overleving als continue hormoontherapie ADT, maar wordt in de praktijk niet vaak toegepast ondanks de voordelen van minder bijwerkingen"