Helpt u ons aan 500 donateurs?
10 december 2018: lees ook dit artikel:
29 maart 2018: Lees ook dit artikel:
29 maart 2018: Bron: Cancer Medicine: 13 March 2018
Ook uit een Aziatische studie blijkt dat Ibrutinib (Imbruvica) superieure resultaten geeft in vergelijking met Rituximab bij recidief van CLL - Chronische Lymfatische Leukemie en SLL, een vorm van lymfklierkanker. (Zie ook in gerelateerde artikelen bv deze studie)
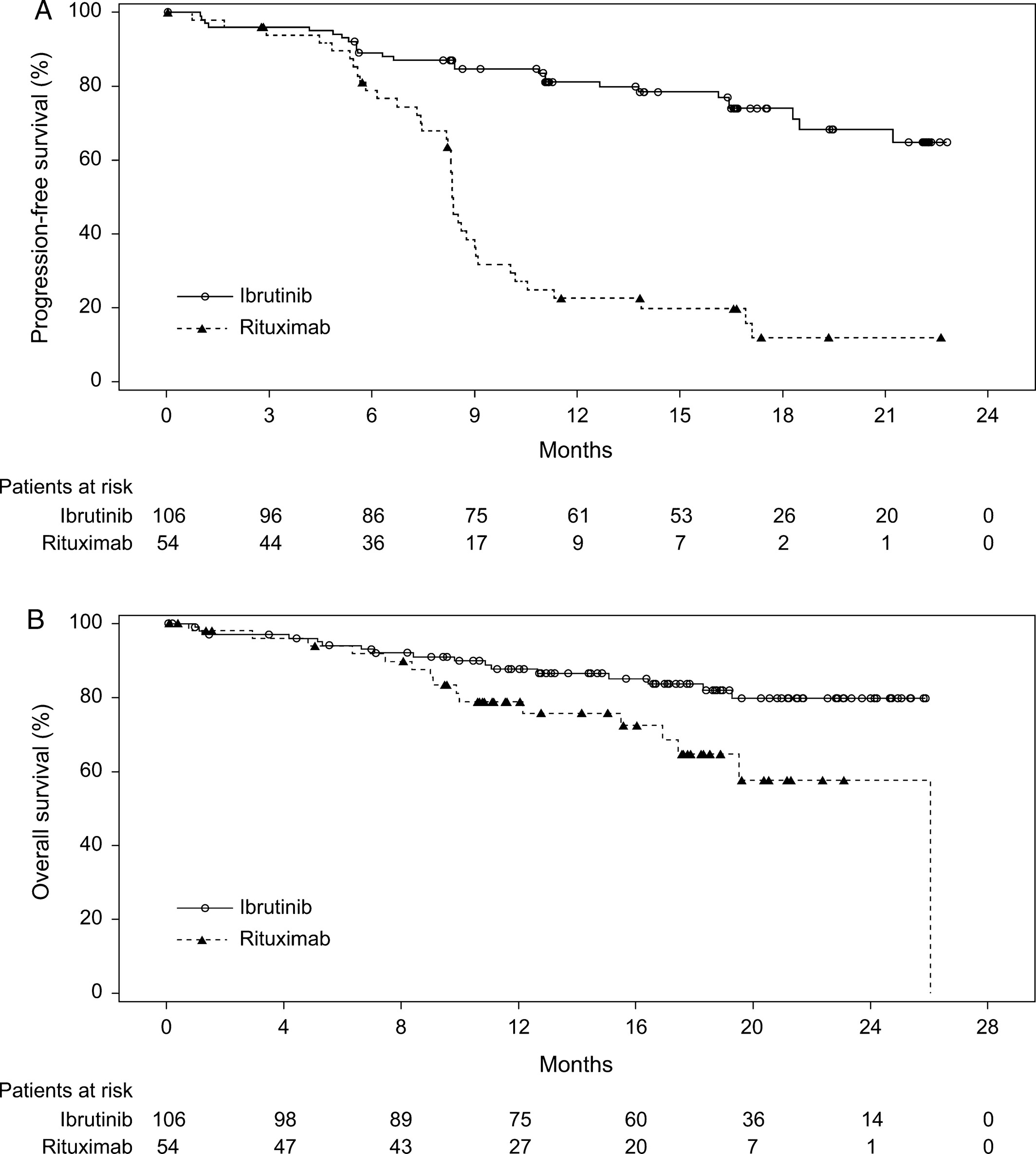
Na een mediane studiefollow-up van 17,8 maanden blijkt de ziektevrije tijd (OOR) al een verschil te geven van plus 45 procent en ook de overall overleving (OS) was al significant beter.
ORR was significantly higher (P < 0.0001) with ibrutinib (53.8%) than with rituximab (7.4%). At a median follow‐up of 17.8 months, ibrutinib improved OS compared with rituximab (HR = 0.446; 95% CI: 0.221–0.900; P = 0.0206).
Hier de grafiek uit het studierapport: Ibrutinib versus rituximab in relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma: a randomized, open‐label phase 3 study
en ik geef maar geen verdere vertaling van de details want deze zijn duidelijk lijkt mij. Ga naar het volledige studierapprot en bekjk alle grafieken enz. Onderaan de grafiek staat het abstract met referentielijst.
Efficacy
In the updated analysis, 64 PFS events were reported (26 [24.5%] in the ibrutinib arm and 38 [70.4%] in the rituximab arm). PFS was significantly improved for patients in the ibrutinib arm compared with the rituximab arm (HR = 0.180, 95% CI: 0.105–0.308; P < 0.0001). The median PFS was not reached in the ibrutinib arm; median PFS for the rituximab arm was 8.3 months (range, 0–22.6 months). At the 18‐month landmark, the estimated PFS rate in the ibrutinib arm was 74.0%, and in the rituximab arm, it was 11.9% (Fig. 1A).
Ibrutinib significantly improved PFS, ORR, and OS compared with rituximab; the results were robust and internally consistent. Ibrutinib displayed a manageable safety profile with no new or unexpected events reported.
Ibrutinib versus rituximab in relapsed or refractory chronic lymphocytic leukemia or small lymphocytic lymphoma: a randomized, open‐label phase 3 study
Abstract
In the Asia‐Pacific region, treatment options are limited for patients with relapsed/refractory chronic lymphocytic leukemia (CLL)/small lymphocytic lymphoma (SLL). Rituximab is widely used in this setting when purine analog‐based therapies are not appropriate. We evaluated the efficacy and safety of ibrutinib compared with rituximab in a randomized, open‐label phase 3 study in predominantly Asian patients with relapsed/refractory CLL/SLL. Patients (N = 160) were randomly assigned 2:1 to receive 420 mg ibrutinib (n = 106) until disease progression (PD) or unacceptable toxicity or up to six cycles of rituximab (n = 54). The primary endpoint was investigator‐assessed progression‐free survival (PFS); key secondary endpoints were overall response rate (ORR), overall survival (OS), and safety. Rituximab‐treated patients could crossover to receive ibrutinib after confirmed PD. At data cutoff, median treatment duration was 16.4 months for ibrutinib and 4.6 months for rituximab. Ibrutinib significantly improved PFS (hazard ratio = 0.180, 95% confidence interval : 0.105–0.308). ORR was significantly higher (P < 0.0001) with ibrutinib (53.8%) than with rituximab (7.4%). At a median follow‐up of 17.8 months, ibrutinib improved OS compared with rituximab (HR = 0.446; 95% CI: 0.221–0.900; P = 0.0206). Overall incidence of adverse events (AEs) was similar between treatments and was not exposure‐adjusted. With ibrutinib, most common AEs were diarrhea and platelet count decreased; with rituximab, most common AEs were neutrophil count decreased and platelet count decreased. Grade ≥3 AEs were reported in 82.7% of ibrutinib‐treated patients and 59.6% of rituximab‐treated patients. Ibrutinib improved PFS, ORR, and OS compared with rituximab and displayed a manageable safety profile in Asian patients with relapsed/refractory CLL/SLL.
References:
- 1Eichhorst, B., T. Robak, E. Montserrat, P. Ghia, P. Hillmen, M. Hallek, et al. 2015. Chronic lymphocytic leukaemia: ESMO clinical practice guidelines for diagnosis, treatment and follow‐up. Ann. Oncol. 26(Suppl. 5):v78–v84.
- 2Chihara, D., H. Ito, T. Matsuda, A. Shibata, A. Katsumi, S. Nakamura, et al. 2014. Differences in incidence and trends of haematological malignancies in Japan and the United States. Br. J. Haematol. 164:536–545.
- 3Redaelli, A., B. L. Laskin, J. M. Stephens, M. F. Botteman, and C. L. Pashos. 2004. The clinical and epidemiological burden of chronic lymphocytic leukaemia. Eur. J. Cancer Care (Engl.) 13:279–287.
- 4Yang, S. M., J. Y. Li, R. P. Gale, and X. J. Huang. 2015. The mystery of chronic lymphocytic leukemia (CLL): why is it absent in Asians and what does this tell us about etiology, pathogenesis and biology? Blood Rev. 29:205–213.
- 5Yoon, S. O., C. Suh, D. H. Lee, H. S. Chi, C. J. Park, S. S. Jang, et al. 2010. Distribution of lymphoid neoplasms in the Republic of Korea: analysis of 5318 cases according to the World Health Organization classification. Am. J. Hematol. 85:760–764.
- 6Gale, R. P., W. Cozen, M. T. Goodman, F. F. Wang, and L. Bernstein. 2000. Decreased chronic lymphocytic leukemia incidence in Asians in Los Angeles County. Leuk. Res. 24:665–669.
- 7Mak, V., D. Ip, O. Mang, C. Dalal, S. Huang, A. Gerrie, et al. 2014. Preservation of lower incidence of chronic lymphocytic leukemia in Chinese residents in British Columbia: a 26‐year survey from 1983 to 2008. Leuk. Lymphoma 55:824–827.
- 8Chan, T. S., Y. S. Lee, I. Del Giudice, M. Marinelli, C. Ilari, L. Cafforio, et al. 2017. Clinicopathological features and outcome of chronic lymphocytic leukaemia in Chinese patients. Oncotarget 8:25455–25468.
- 9Dong, H. J., L. T. Zhou, D. X. Zhu, D. M. Wang, C. Fang, H. Y. Zhu, et al. 2011. The prognostic significance of TP53 mutations in Chinese patients with chronic lymphocytic leukemia is independent of del(17p13). Ann. Hematol. 90:709–717.
- 10Lai, Y. Y., and X. J. Huang. 2011. Cytogenetic characteristics of B cell chronic lymphocytic leukemia in 275 Chinese patients by fluorescence in situ hybridization: a multicenter study. Chin. Med. J. (Engl.) 124:2417–2422.
- 11Qiu, H. X., W. Xu, X. S. Cao, M. Zhou, Y. F. Shen, Y. L. Xu, et al. 2008. Cytogenetic characterisation in Chinese patients with chronic lymphocytic leukemia: a prospective, multicenter study on 143 cases analysed with interphase fluorescence in situ hybridisation. Leuk. Lymphoma 49:1887–1892.
- 12Marinelli, M., C. Ilari, Y. Xia, I. Del Giudice, L. Cafforio, I. Della Starza, et al. 2016. Immunoglobulin gene rearrangements in Chinese and Italian patients with chronic lymphocytic leukemia. Oncotarget 7:20520–20531.
- 13Jang, M. A., E. H. Yoo, K. Kim, W. S. Kim, C. W. Jung, S. H. Kim, et al. 2013. Chronic lymphocytic leukemia in Korean patients: frequent atypical immunophenotype and relatively aggressive clinical behavior. Int. J. Hematol. 97:403–408.
- 14Tomomatsu, J., Y. Isobe, K. Oshimi, Y. Tabe, K. Ishii, M. Noguchi, et al. 2010. Chronic lymphocytic leukemia in a Japanese population: varied immunophenotypic profile, distinctive usage of frequently mutated IGH gene, and indolent clinical behavior. Leuk. Lymphoma 51:2230–2239.
- 15Li, Z., F. Li, S. Yi, Z. Gu, Z. Yu, Y. Xu, et al. 2015. Superior efficacy of rituximab‐based chemoimmunotherapy as an initial therapy in newly diagnosed patients with B cell indolent lymphomas: long‐term results from a single center in China. BMC Cancer 15:555.
- 16Ogawa, Y., M. Ogura, T. Suzuki, K. Ando, T. Uchida, Y. Shirasugi, et al. 2013. A phase I/II study of ofatumumab (GSK1841157) in Japanese and Korean patients with relapsed or refractory B‐cell chronic lymphocytic leukemia. Int. J. Hematol. 98:164–170.
- 17Tobinai, K., M. Ogura, K. Ishizawa, T. Suzuki, W. Munakata, T. Uchida, et al. 2016. Safety and tolerability of ibrutinib monotherapy in Japanese patients with relapsed/refractory B cell malignancies. Int. J. Hematol. 103:86–94.
- 18Wierda, W. G., A. D. Zelenetz, L. I. Gordon, J. S. Abramson, R. H. Advani, C. B. Andreadis, et al. 2017. NCCN guidelines insights: chronic lymphocytic leukemia/small lymphocytic leukemia, version 1.2017. J. Natl. Compr. Canc. Netw. 15:293–311.
- 19Lamanna, N. 2012. Treatment of older patients with chronic lymphocytic leukemia. Curr. Hematol. Malig. Rep. 7:21–25.
- 20Fischer, K., P. Cramer, R. Busch, S. Bottcher, J. Bahlo, J. Schubert, et al. 2012. Bendamustine in combination with rituximab for previously untreated patients with chronic lymphocytic leukemia: a multicenter phase II trial of the German Chronic Lymphocytic Leukemia Study Group. J. Clin. Oncol. 30:3209–3216.
- 21Hallek, M., K. Fischer, G. Fingerle‐Rowson, A. M. Fink, R. Busch, J. Mayer, et al. 2010. Addition of rituximab to fludarabine and cyclophosphamide in patients with chronic lymphocytic leukaemia: a randomised, open‐label, phase 3 trial. Lancet 376:1164–1174.
- 22Buggy, J. J., and L. Elias. 2012. Bruton tyrosine kinase (BTK) and its role in B‐cell malignancy. Int. Rev. Immunol. 31:119–132.
- 23Janssen‐Cilag International NV. 2016. Imbruvica [summary of product characteristics]. Janssen‐Cilag International NV, Beerse, Belgium.
- 24Pharmacyclics LLC. 2017. Imbruvica [package insert]. Pharmacyclics LLC, Sunnyvale, CA.
- 25Byrd, J. C., R. R. Furman, S. E. Coutre, I. W. Flinn, J. A. Burger, K. A. Blum, et al. 2013. Targeting BTK with ibrutinib in relapsed chronic lymphocytic leukemia. N. Engl. J. Med. 369:32–42.
- 26Byrd, J. C., J. R. Brown, S. O'Brien, J. C. Barrientos, N. E. Kay, N. M. Reddy, et al. 2014. Ibrutinib versus ofatumumab in previously treated chronic lymphoid leukemia. N. Engl. J. Med. 371:213–223.
- 27Weiner, G. J. 2010. Rituximab: mechanism of action. Semin. Hematol. 47:115–123.
- 28Furman, R. R., J. P. Sharman, S. E. Coutre, B. D. Cheson, J. M. Pagel, P. Hillmen, et al. 2014. Idelalisib and rituximab in relapsed chronic lymphocytic leukemia. N. Engl. J. Med. 370:997–1007.
- 29Mato, A. R., J. Jahnke, P. Li, M. Mehra, V. P. Ladage, M. Mahler, et al. 2016. Characterization of real world survival outcomes among older adults with chronic lymphocytic leukemia receiving second line treatment in the pre novel‐agents era: an analysis of the 2007‐2013 SEER‐Medicare database. Blood 128:4790.
- 30Ysebaert, L., B. P. Philip, and S. Stilgenbauer. 2014. Real‐world treatment patterns of rituximab usage as single‐agent therapy or part of combination regimens in chronic lymphocytic leukemia (CLL) in Eu5 countries (UK, France, Germany, Italy, and Spain). Value Health 17:A234.
- 31Feinberg, B., B. Schenkel, A. McBride, L. Ellis, and J. Radtchenko. 2015. Real‐world treatment patterns in first line and relapsed chronic lymphocytic leukemia (CLL). Clin. Lymphoma Myeloma Leuk. 15:S204–S205.
- 32Mato, A., C. Nabhan, N. E. Kay, M. A. Weiss, N. Lamanna, T. J. Kipps, et al. 2016. Real‐world clinical experience in the Connect® chronic lymphocytic leukaemia registry: a prospective cohort study of 1494 patients across 199 US centres. Br. J. Haematol. 175:892–903.
- 33Satram‐Hoang, S., C. Reyes, K. Q. Hoang, F. Momin, and S. Skettino. 2014. Treatment practice in the elderly patient with chronic lymphocytic leukemia‐analysis of the combined SEER and Medicare database. Ann. Hematol. 93:1335–1344.
- 34Hallek, M., B. D. Cheson, D. Catovsky, F. Caligaris‐Cappio, G. Dighiero, H. Dohner, et al. 2008. Guidelines for the diagnosis and treatment of chronic lymphocytic leukemia: a report from the International Workshop on Chronic Lymphocytic Leukemia updating the National Cancer Institute‐Working Group 1996 guidelines. Blood 111:5446–5456.
- 35Hallek, M., B. D. Cheson, D. Catovsky, F. Caligaris‐Cappio, G. Dighiero, H. Dohner, et al. 2012. Response assessment in chronic lymphocytic leukemia treated with novel agents causing an increase of peripheral blood lymphocytes. Blood http://www.bloodjournal.org/content/111/12/5446/tab-e-letters.
- 36Marostica, E., J. Sukbuntherng, D. Loury, J. de Jong, X. W. de Trixhe, A. Vermeulen, et al. 2015. Population pharmacokinetic model of ibrutinib, a Bruton tyrosine kinase inhibitor, in patients with B cell malignancies. Cancer Chemother. Pharmacol. 75:111–121.
- 37Brown, J., P. Hillmen, S. O'Brien, J. Barrientos, N. Reddy, S. Coutre, et al. 2017. Extended follow‐up and impact of high‐risk prognostic factors from the phase 3 RESONATE™ study in patients with previously treated CLL/SLL. Leukemia. https://doi.org/10.1038/leu.2017.175.
- 38Byrd, J. C., R. R. Furman, S. E. Coutre, J. A. Burger, K. A. Blum, M. Coleman, et al. 2015. Three‐year follow‐up of treatment‐naive and previously treated patients with CLL and SLL receiving single‐agent ibrutinib. Blood 125:2497–2506.
- 39Huhn, D., C. von Schilling, M. Wilhelm, A. D. Ho, M. Hallek, R. Kuse, et al. 2001. Rituximab therapy of patients with B‐cell chronic lymphocytic leukemia. Blood 98:1326–1331.
- 40Itala, M., C. H. Geisler, E. Kimby, E. Juvonen, G. Tjonnfjord, K. Karlsson, et al. 2002. Standard‐dose anti‐CD20 antibody rituximab has efficacy in chronic lymphocytic leukaemia: results from a Nordic multicentre study. Eur. J. Haematol. 69:129–134.
- 41Nguyen, D. T., J. A. Amess, H. Doughty, L. Hendry, and L. W. Diamond. 1999. IDEC‐C2B8 anti‐CD20 (rituximab) immunotherapy in patients with low‐grade non‐Hodgkin's lymphoma and lymphoproliferative disorders: evaluation of response on 48 patients. Eur. J. Haematol. 62:76–82.
- 42Winkler, U., M. Jensen, O. Manzke, H. Schulz, V. Diehl, and A. Engert. 1999. Cytokine‐release syndrome in patients with B‐cell chronic lymphocytic leukemia and high lymphocyte counts after treatment with an anti‐CD20 monoclonal antibody (rituximab, IDEC‐C2B8). Blood 94:2217–2224.
- 43Burger, J. A., A. Tedeschi, P. M. Barr, T. Robak, C. Owen, P. Ghia, et al. 2015. Ibrutinib as initial therapy for patients with chronic lymphocytic leukemia. N. Engl. J. Med. 373:2425–2437.
- 44Shanafelt, T. D., S. A. Parikh, P. A. Noseworthy, V. Goede, K. G. Chaffee, J. Bahlo, et al. 2017. Atrial fibrillation in patients with chronic lymphocytic leukemia (CLL). Leuk. Lymphoma 58:1630–1639.
Gerelateerde artikelen
- Pirtobrutinib, een niet-covalente BTK remmer, blijkt effectief bij zwaar voorbehandelde patiënten met chronische lymfatische leukemie (CLL) of kleincellig lymfatisch lymfoom (SLL) die eerder een covalente BTK remmer hadden gehad
- Reviewstudie beschrijft hoe je het beste patienten met CLL = Chronische Lymfatische Leukemie kunt diagnosteren en welke behandelingen het beste zijn te gebruiken in verschillende stadia van de ziekte.
- Venetoclax plus rituximab geeft ook bij jonge en fitte patiënten met chronische lymfatische leukemie en zelfs bij ongemuteerde IGHV veel complete remissies
- acalabrutinib als monotherapie verbeterde progressievrije overleving aanzienlijk met lager bijwerkingenprofiel dan idelalisib + rituximab of bendamustine + rituximab bij gevorderde CLL - chronische lymfatische leukemie
- Ibrutinib geeft veel betere resultaten op ziekteprogressievrije overleving (87 vs 74 procent) dan bendamustine plus rituximab bij onbehandelde oudere CLL patienten. Rituximab lijkt zinloos naast ibrutinib
- Man - 48 jaar - met al 20 jaar Chronische Lymfatische Leukemie (CLL) weigert chemo en bereikt een langdurige en duurzame remissie na behandeling met Epigallocatechin-3-gallate, een extract van groene thee. copy 1
- BCL-2 remmer - ABT-199/GDC-0199 geeft spectaculaire resultaten bij vergevorderde zwaar voorbehandelde CLL -Chronische Lymfatische Leukemie met alsnog 23 procent totale remissies van minimaal 2 jaar
- Ibrutinib geeft veel betere ziektevrije tijd (plus 45 procent) in vergelijking met rituximab bij recidief van chronische lymfatische leukemie en SLL - lymfklierkanker
- Ibrutinib aanvullend op Bendamustine en Rituximab verbetert progressievrije ziekte en overall overleving met tientallen procenten bij Chronische Lymfatische Leukemia en recidief van lymfklierkanker met kleine tumoren copy 1
- Ibrutinib - Tyrosine kinase (BTK) remmer PCI 32.765 - Ibrutinib zorgt voor langere ziektevrije tijd met veel minder bijwerkingen bij patiënten met CLL - chronische lymfatische leukemie bevestigen nieuwe studies copy 1
- Idelalisib naast rituximab zorgt voor significant langere overleving en progressievrije tijd bij patienten met CLL - chronische lymfatische leukemie waarvoor chemo niet meer mogelijk was
- Groene thee extract zorgt binnen enkele maanden voor significante remissie bij CLL patienten (Chronische Lymfatische Leukemie) . Nieuwe fase II studie bevestigt eerdere goede resultaten. copy 1
- Ofatumumab door FDA goedgekeurd als medicijn voor patienten met CLL - Chronische Lymfatische Leukemie, ongevoelig geworden voor fludarabine en alemtuzumab.
- Combinatiebehandeling van fludarabine en alemtuzumab geeft significant langere ziektevrije tijd en betere overleving voor eerder behandelde CLL patienten
- Rituximab naast Fludarabine en cyclophosphamide geeft significant betere resultaten op ziektevrije tijd en op overleving bij CLL en wordt eerste lijns.
- CLL - Chronische Lymfatische Leukemie: een overzicht




Plaats een reactie ...
Reageer op "Ibrutinib geeft veel betere ziektevrije tijd (plus 45 procent) in vergelijking met rituximab bij recidief van chronische lymfatische leukemie en SLL - lymfklierkanker"