17 maart 2021: Aanvullend op onderstaande informatie hier het volledige studierapport Low-dose cyclophosphamide selectively expands resident anti-tumor Tcells allowing in situ control of colorectal cancer in PDF formaat.
28 maart 2017 lees ook dit artikel:
28 maart 2017. Bron: ASCO-SITC (Society for Immunotherapy of Cancer) Clinical Immuno-Oncology Symposium
Immuuntherapie met een gemoduleerd virus, het zogeheten Ankara-ST4 virus, plus een lage dosis cyclophosphamide geeft uitstekende resultaten bij eerder met chemo voorbehandelde inoperabele darmkankerpatienten met vergevorderde darmkanker. Progressievrije ziektetijd ging van 2,4 naar 5 maanden en mediane overall overleving van 11,2 naar 20,0 maanden in de groep patiënten met de optimale behandeling.
Dit blijkt uit een kleinschalige open-label fase I/II veiligheidsstudie bij totaal 52 patienten met inoperabele darmkanker.
Bijna alle patiënten hadden eerder een chemokuur gehad met of capecitabine (Xeloda) en 5-FU (fluorouracil) en de meesten hadden daarna ook nog andere chemokuren of behandelingen gehad.
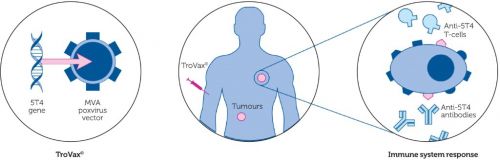 Werkingsmechanisme van het Ankara virus (TroVax)
Werkingsmechanisme van het Ankara virus (TroVax)
Studie opzet:
Patiënten werden gerandomiseerd ingedeeld in geen behandeling (n = 8), of een lage dosis cyclophosphamide met 50 mg 2x daags gedurende behandelingsweken 1 en 3 (n = 9), of een vaccin met het gemoduleerde virus Ankara–5T4 only (n = 17), of lage dosis cyclophosphamide gevolgd door het gemoduleerde virus Ankara–5T4 (n = 18). Patiënten in de vaccingroep startten de behandeling op dag 22, nadat zij eerder cyclophosphamide hadden gekregen. Zij kregen 5 injecties om de 2 weken de 6e injectie 4 weken na de 5e injectie. Het primaire doel was te zien of de behandeling een boost te zien gaf in de anti-5T4 genen op dag 43 door het meten van gestegen T-cellen en/of antibody responses.
Kernpunten uit de studie:
- A novel therapeutic vaccine employs a highly attenuated strain of vaccinia virus, modified vaccinia virus Ankara, and encodes the tumor antigen 5T4, which is found on 90% of colorectal cancers.
- In a phase I/II trial of advanced colorectal cancer patients, modified vaccinia virus Ankara–5T4 plus low-dose cyclophosphamide (delivered prior to vaccination) led to robust immune responses that were associated with improved progression-free and overall survival.
- Low-dose cyclophosphamide alone also produced strong immune responses that were associated with prolonged remission.
Een opmerkelijk citaat van de studieleider:
This is the first randomized study to show a clear benefit of immunotherapy in advanced colorectal cancer—and to suggest this approach may be superior to (and less toxic than) continuous palliative chemotherapy in these patients.
Studie resultaten:
“Nadat we de cyclofosfamide gaven, zagen we dat de patiënten met de grootste vermindering van regulerende onderdrukkende T-cellen het meeste profiteerden met een progressievrije overleving,” aldus Dr. Scurr gemeld. “Gewoon het geven van een lage dosis cyclofosfamide alleen gaf ook al een immuunrespons te zien.”
Cyclophosphamide verzwakte regulerende onderdrukkende T cellen bij 21 van de 27 patiënten gedurende de eerste drie weken (P = .0028). Onder de 27 met cyclophosphamide behandelde patiënten, bereikten er 12 een vermindering van minimaal 39.4% (het doel dat statistisch significantie betekende). Deze verzwakking van de regulerende T-cellen werd ook gerelateerd aan een langere progressie vrije ziekte in de groepen die ook cyclophosphamide hadden gekregen, vergeleken met de groep patiënten die niet de 39,4% vermindering hadden gehaald. Mediane progressie vrije ziekte werd 5.0 vs 2.4 maanden voor deze groep van patiënten. (hazard ratio = 0.48, P = .09).
Een vaccinatie erbij verdubbelde de anti-5T4 immuun reacties bij 16 van de 35 patiënten welke waren behandeld met het gemoduleerde virus Ankara–5T4. Deze patiënten ervaarden een progressievrije ziekte van 5,6 maanden versus 2,4 maanden. (HR = 0.21, P = .0002) en een mediane overall overleving van 20.0 maanden vs 11.2 maanden; HR = 0.32, P = .0076).
Belangrijk te melden dat er geen extra ernstige bijwerkingen optraden bij deze aanpak.
In dit studie rapport, vrij in te zien: TroVax in colorectal cancer wordt beschreven hoe TROVAX werkt en gaat vergezeld van een interessante referentielijst. Zie ook hieronder. Interessant is ook dat Trovax bij ook andere vormen van kanker b.v. nierkanker, prostaatkanker wordt onderzocht en hoopvolle resultaten geeft.
Een andere studie van immuuntherapie met het gemoduleerde virus Ankara is deze review studie: 5T4-modified vaccinia Ankara: progress in tumor-associated antigen-based immunotherapy maar daarvoor moet worden betaald. En het abstract geeft te weinig informatie om daaruit conclusies op te kunnen maken.
Het studierapport: Scurr M, Pembroke T, Adams R et al: MVA-5T4 immunotherapy and low-dose cyclophosphamide for advanced colorectal cancer (TaCTiCC): An open-label, randomized phase I/II trial is nog niet vrij in te zien.
Hieronder wel het abstract na het abstract en referentielijst van de eerder genoemde studie: TroVax in colorectal cancer
The existing results support the ability of TroVax to induce immune responses within the host;
TroVax in colorectal cancer
Conclusions: Both CPM and TroVax induced highly beneficial anti-tumor immune responses resulting in significantly prolonged survival of end-stage CRC patients without toxicity. This is the first study to show a clear benefit of immunotherapy in advanced CRC, and suggests this approach may be superior (and less toxic) to continuous palliative chemotherapy in these patients.
Therapies Targeting T cells
154
Background: Current immunotherapies including checkpoint inhibitors and vaccines for advanced colorectal cancer (CRC) have been largely ineffective. We hypothesized that combining an MVA-based vaccine targeting the tumor-associated antigen 5T4 (TroVax) with low-dose cyclophosphamide to deplete Foxp3+regulatory T-cells (Tregs), could improve immunological responses and patient outcomes.
Methods: In this open-label phase I/II clinical trial, TaCTiCC (TroVax and Cyclophosphamide Treatment in Colorectal Cancer) 53 patients with inoperable metastatic CRC were randomized to receive either no treatment (group 1, n=8), metronomic low-dose CPM (50mg B.D. during treatment weeks 1&3; group 2, n=9), TroVax only (6 i.m. injections weeks 4 to 16, group 3, n=18), or low-dose CPM followed by TroVax (group 4, n=18). The primary endpoint was boosted anti-5T4 responses at week 7, as measured by increased T-cell and antibody responses; secondary endpoints included progression-free (PFS)/overall survival (OS), and anti-5T4 responses over the trial period.
Results: CPM depleted Tregs in 21/27 patients during treatment week 3 (p=0.0045), resulting in significantly prolonged PFS amongst groups 2&4 over group 1 (5.0 vs. 2.5 months, HR=0.17 95% CI 0.048-0.62, p=0.0072). TroVax induced a >2-fold increase in anti-5T4 immune responses in 15/36 group 3&4 patients; these patients experienced significantly prolonged median PFS (6.5 vs. 2.4 months, HR 0.31 95% CI 0.14-0.65, p=0.0022) and OS (20 vs. 12 months, HR=0.37 95% CI 0.17-0.82, p=0.014). Combination of CPM & TroVax was not significantly superior. The primary endpoint at a single timepoint was not met since CPM-induced responses declined by week 7, and TroVax-induced responses were greatest at weeks 10-16. No serious adverse events were reported.
Conclusions: Both CPM and TroVax induced highly beneficial anti-tumor immune responses resulting in significantly prolonged survival of end-stage CRC patients without toxicity. This is the first study to show a clear benefit of immunotherapy in advanced CRC, and suggests this approach may be superior (and less toxic) to continuous palliative chemotherapy in these patients. Clinical trial information: 54669986.
Gerelateerde artikelen
- Maag-darmkanker: Zanidatamab in combinatie met chemotherapie, al dan niet aangevuld met tislelizumab, geeft uitstekende resultaten bij patiënten met HER2-positieve, gevorderde of uitgezaaide maag- en slokdarmkanker copy 1
- Trastuzumab plus Tucatinib gericht op HER2 expressie vooraf aan chemo voorkomt belastende operatie als eerstelijns behandeling bij patienten met onbehandelde rectumkanker met betere kwaliteit van leven omdat operatie niet meer nodig was
- Nous-209, gepersonaliseerde immuungerichte vaccins tonen veelbelovende resultaten bij het voorkomen van erfelijke kanker bij patiënten met het Lynchsyndroom, waaronder blaaskanker, buikvlieskanker, darmkanker, longkanker copy 1
- crispr-cas9-bewerkte T-cellen gericht op CISH maakt patienten met gevorderde darmkanker en GIST alsnog gevoelig voor immuuntherapie met anti-PD medicijnen
- Immuuntherapie met nivolumab plus ipilimumab vooraf aan operatie bij darmkankerpatienten met hoge waarden van microsatellite-instability high/mismatch repair deficient (MSI-H/dMMR) blijkt zeer effectief.
- Immuuntherapie met Pembrolizumab geeft veel betere resultaten op ziektevrije overleving (48 vs 18 procent op 2 jaars meting) dan chemotherapie voor uitgezaaide darmkanker met MSI-H/dMMR - of afwijkende reparatie genen
- KRAS gemuteerde tumoren: Kankervaccin ELI-002 2P stimuleerde hoge T-celreacties bij patiënten met voor immuuntherapie ongevoelige KRAS-gemuteerde tumoren en verbeterde de ziekteprogressieve tijd bij patienten met alvleesklierkanker en darmkanker copy 1 co
- Immuuntherapie vooraf aan operatie en chemotherapie blijkt succesvol bij kankerpatiënten met maagkanker en met tumoren op de overgang van slokdarm naar de maag copy 2
- Dostarlimab, een specifieke vorm van een anti-PD medicijn is 100 procent effectief bij alle 12 patienten met operabele rectumkanker met dMMR = Mismatch-reparatie-deficiëntie en was geen operatie meer nodig.
- Kankerpatienten met solide tumoren met MSI-H = hoge microsatelliet instabiliteit en mismatch reparatie (dMMR) reageren uitstekend op immuuntherapie met pembrolizumab vooraf aan operatie met 65 tot 80 procent complete remissies copy 1
- Immuuntherapie met nivolumab plus ipilimuab voorafgaand aan operatie darmkanker geeft uitstekende resultaten met duurzame klinische complete remissies
- Temozolomide - temodal gevolgd door immunotherapie met combinatie van lage dosis ipilimumab plus nivolumab geeft hoopgevende resultaten bij patiënten met microsatellietstabiel en MGMT-gedempte uitgezaaide darmkanker
- CYAD-101, een vorm van CAR-T cel immuuntherapie geeft hoopvolle resultaten bij uitgezaaide darmkanker zonder dat graft-versus-host-ziekte ontstaat.
- Dendritische cellen en Newcastle Disease Virus bij kankerpatiënten met spijsverteringskanker geeft significant betere resultaten in overlevingstijd aldus gerandomiseerde studie bij 335 patiënten. copy 2
- Autologe genetisch gemodificeerde T-cellen gericht tegen het humaan papillomavirus (HPV) 16 E6 geeft bij patienten met vergevorderde zwaar voorbehandelde uitgezaaide HPV gerelateerde kanker uitstekende resultaten copy 1
- Man met uitgezaaide darmkanker stadium 4 (KRAS pos.) geneest alsnog met combinatie van immuuntherapie met Rigvir virus (dendritische celtherapie) plus FOLFOX en bevacizumab en is nu na 8 jaar kankervrij.
- Nivolumab (Opvido) + ipilimumab (Yervoy) geeft uitstekende resultaten bij nog niet behandelde uitgezaaide darmkanker (met MSI-H of dMMR mutaties) en bereikte 53 procent een gedeeltelijke remissie en 7 procent een complete remissie.
- Immuuntherapie met een gemoduleerd virus plus avelumab, een anti-PD medicijn, wordt onderzocht in een fase I/II studie bij darmkankerpatienten
- Man met uitgezaaide inoperabele darmkanker komt met ATID - Autologous tumor immunizing devascularization, een vorm van immuuntherapie in complete remissie en is al 16 jaar kankervrij, zonder chemo of andere behandelingen
- Immuuntherapie met autovaccinatie van dendritische cellen moet weer eerstelijns behandeling worden voor operabele darmkanker stadium II en III
- Immuuntherapie met dendritische celtherapie geeft uitstekende resultaten op overall overleving en ziektevrije tijd bij darmkanker met weinig of geen zichtbare tumoren.
- Xilonix - MAPp1 zorgt voor stabiele ziekte (bij 53 procent) bij zwaar voorbehandelde darmkankerpatienten stadium 4 met een mediane overall overleving van 4.2 maanden vs 11.5 maanden in vergelijking met placebo copy 1
- Dendritische cellen en Newcastle Disease Virus bij kankerpatiënten met spijsverteringskanker geeft significant betere resultaten in overlevingstijd aldus gerandomiseerde studie bij 335 patiënten. copy 1
- Immuuntherapie: Oncophage(R) (HSPPC-96), gemaakt van eigen tumorcellen zorgt voor opmerkelijke resultaten bij o.a. darmkanker.
- Immuuntherapie met het gemoduleerde virus Ankara–5T4 (TroVax) plus lage dosis cyclophosphamide zorgt voor verdubbeling van mediane overall overleving 11,2 vs 20 maanden bij vergevorderde darmkanker
- Immuuntherapie geeft uitstekende resultaten bij darmkanker met minimale ziekte - weinig tumoren - en zelfs bij darmkanker stadium IV werkt het ook al is het dan minder effectief
- Immuuntherapie met een moleculair middel - codenaam MGN1703 - geeft hoog significant betere ziektevrije tijd bij patiënten met uitgezaaide darmkanker die eerder chemotherapie kregen.
- Autovaccinatie bij darmkanker. Oncovax geeft significant beter resultaat op overleving, overlevingstijd en tijd tot recidief bij darmkankerpatiënten met stadum II. Intracel is failliet verklaard. Oncovax wordt niet meer geleverd
- Archief: immuuntherapie met CEA anti-body plus bestraling geeft hoopvolle resultaten in Phase-II trial met 30 darmkankerpatiënten.
- Immuuntherapie waaronder dendritische celtherapie bij vormen van darmkanker, een overzicht



Plaats een reactie ...
Reageer op "Immuuntherapie met het gemoduleerde virus Ankara–5T4 (TroVax) plus lage dosis cyclophosphamide zorgt voor verdubbeling van mediane overall overleving 11,2 vs 20 maanden bij vergevorderde darmkanker"