Mocht u de informatie op onze website kanker-actueel.nl waarderen dan wilt u ons misschien ondersteunen met een donatie?
Ons rekeningnummer is: RABO 37.29.31.138 t.n.v. Stichting Gezondheid Actueel in Terneuzen.
Onze IBANcode is NL79 RABO 0372 9311 38
Als donateur kunt u ook korting krijgen bij verschillende bedrijven. En we hebben een ANBI status
Zie ook in gerelateerde artikelen hiernaast of hieronder.14 april 2026: Bron: Naturer 25 maart 2026
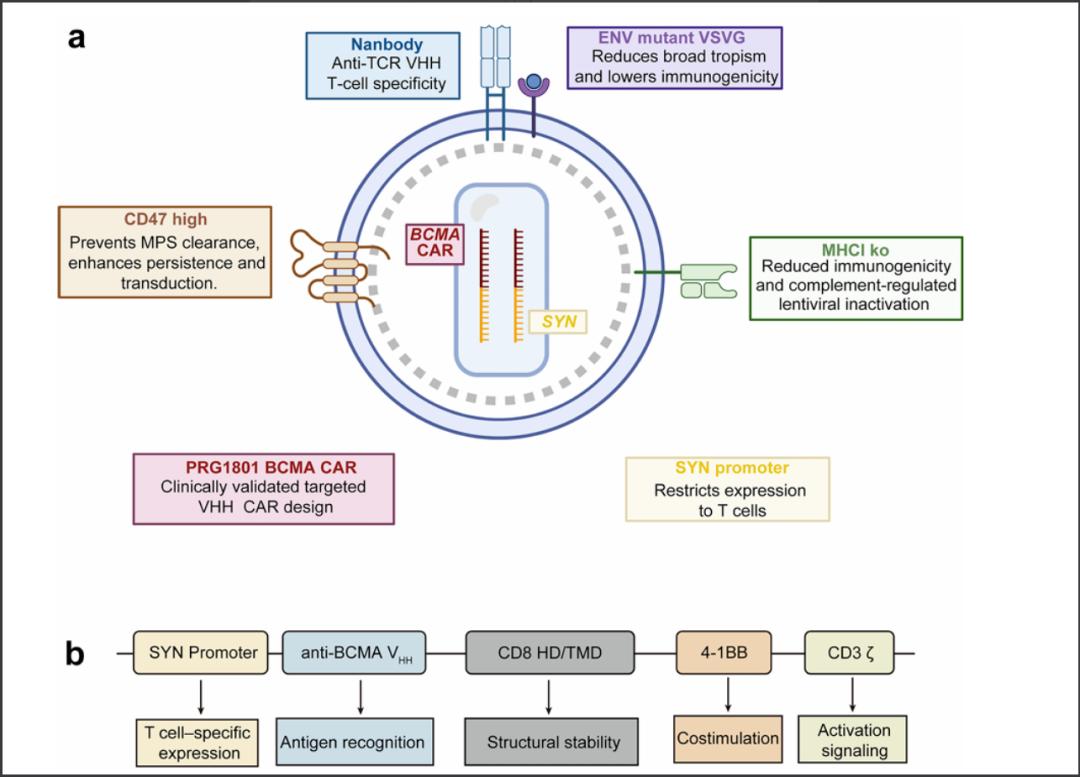
Vijf zwaar voorbehandelde patiënten met een recidief of ziekteprogressie van uitgezaaide Multiple Myeloma (botkanker - ziekte van Kahler) boekten uitstekende resultaten door een eenmalige intraveneuze infusie van ESO-T01 – een zogeheten nanobody-gerichte, immuun afgeschermde lentivirale vector die zich richt op de receptor BCMA CAR. Bijzonder aan deze studie is dat er voor de productie van ESO-T01 vooraf geen leukafarese of chemotherapie nodig was. ESO-T01 richtte zich selectief op in bloed circulerende T-cellen en herprogrammeerde deze in het lichaam om het CAR-T medicijn tot expressie te brengen, waarna deze cellen zich vermenigvuldigden en een sterke antitumoractiviteit vertoonden zonder gezonde cellen aan te vallen. Blijkt uit een eerste verkennende studie bij 5 patiënten.
Ondanks de kleine studiegroep van slechts 5 patiënten was de effectiviteit wel opvallend positief: 4 van de 5 patiënten bereikten objectieve remissies, waaronder drie volledige aangetoonde complete remissies, met een restziekte (MRD) -negativiteit bij een gevoeligheid van 10⁻⁵ bij alle vier de evalueerbare patiënten op dag 60.
Het Cytokine-release-syndroom als ernstige bijwerking trad op bij 80% van de patiënten (3 patiënten hadden graad 3, 1 patiënt graad 2), die wel allemaal onder controle te krijgen waren met een standaardbehandeling. De meest voorkomende bijwerkingen waren voorbijgaande cytopenie en een behandelbare verhoging van de leverenzymen. Tijdens de studie overleed één patiënt als gevolg van ruggenmergcompressie veroorzaakt door extramedullaire laesies, wat nauw verband hield met de ziekteprogressie.
Hier een grafiek uit het studierapport gekopieerd
Hoewel de duurzaamheid onzeker blijft – de mediane follow-up was beperkt tot 6 maanden en de studie werd voortijdig beëindigd in 2025 – is het biologische bewijs van het concept overtuigend en heeft het implicaties die veel verder reiken dan deze eerste verkennende studie aldus de onderzoekers.
Al meer dan tien jaar wordt het verandering brengende potentieel van CAR-T-therapie beperkt door een fundamenteel knelpunt: de productie van de CAR-T cellen duurt lang en is erg duur. De benodigde leukafarese, gecentraliseerde productietijdlijnen, chemotherapie om eventueel aangetaste lymfklieren te behandelen en monitoring in het ziekenhuis leiden gezamenlijk tot vertragingen, kosten en klinische risico's, met name voor patiënten met een agressieve vorm van de ziekte die al vóór de infusie kunnen verslechteren.
Tegen deze achtergrond vertegenwoordigt deze studie geen stapsgewijze behandelingsverbetering, maar een verschuiving in produceren en toepassen van een behandeling: de in vivo generatie / ontwikkelen en produceren van CAR-T-cellen, waarbij het traditionele productieproces volledig wordt omzeild. Met als gevolg dat patiënten die hiervoor in aanmerking komen veel eerder een behandeling kunnen krijgen.
Het volledige studierapport is tegen betaling in te zien en gepubliceerd in Nature:
- Article
- Published:
In vivo generation of anti-BCMA CAR-T cells in relapsed or refractory multiple myeloma: a phase 1 study
Nature Medicine (2026)
Abstract
In vivo chimeric antigen receptor (CAR)-T cell generation can bypass ex vivo manufacturing and lymphodepletion, potentially simplifying and accelerating access to cellular therapy; preliminary clinical experience supports feasibility and suggests preliminary efficacy. This phase 1, single-arm, open-label trial evaluated the safety and tolerability of ESO-T01, a nanobody-directed, immune-shielded lentiviral vector encoding a humanized anti-B cell maturation antigen (BCMA) CAR, in adults with relapsed or refractory multiple myeloma. ESO-T01 was administered as a single intravenous infusion of 0.2 × 109 transduction units without leukapheresis, ex vivo manufacturing or lymphodepleting chemotherapy. Five heavily pretreated male patients (median three prior lines) were consecutively enrolled and followed for a median of 6.0 months. The trial was stopped early in 2025, and no further enrollment was performed. The primary endpoint was safety and tolerability, and secondary endpoints included efficacy, pharmacokinetics and pharmacodynamics of ESO-T01. No dose-limiting toxicities occurred. All patients developed grade 3 or higher adverse events. Cytokine release syndrome occurred in four patients (three grade 3 and one grade 2) and was managed with corticosteroids, tocilizumab, or supportive care. The most frequent toxicities were transient cytopenias and reversible hepatic enzyme elevations, and three patients experienced grade 2 infections. One patient developed grade 1 immune effector cell-associated neurotoxicity and died from extramedullary lesion-related spinal cord compression. Preliminary antimyeloma activity was observed: four of five patients achieved objective responses, including three stringent complete remissions, with minimal residual disease negativity (10−5) in all evaluable responders (4/4) by day 60. These findings provide preliminary evidence on the feasibility and safety of in vivo CAR-T generation using an immune-shielded vector. ClinicalTrials.gov registration: NCT06791681.
Gerelateerde artikelen
- ESO-T01 toegediend in een eenmalige intraveneuze injectie geeft uitstekende resultaten van deze vorm van CAR-T celtherapie bij patienten met Multiple Myeloma (botkanker - Kahler)
- CAR-T therapie met Ciltacabtagene autoleucel blijkt ook in klinische praktijk uitstekende behandeling met complete remissie van 74 procent voor patienten met recidief of gevorderde multiple Myeloma (Kahler - botkanker)
- Immuuntherapie met anti-BCMA/GPRC5D bispecifieke CAR T-cellen geeft als behandeling bij patiënten met recidief van Multiple Myeloma - Kahler uitstekende resultaten
- CAR T-celtherapie met het B-cell maturation antigen (BCMA) geeft alsnog complete remissies bij meer dan de helft van de 18 geselecteerde zwaar voorbehandelde patienten met vergevorderde multiple myeloma (Kahler)
- JNJ-4528 is een vorm van CAR-T cel behandeling en geeft uitstekende resultaten met 67 procent duurzame complete remissie in nieuwe studie bij uitbehandelde patienten met vergevorderde en zwaar voorbehandelde multiple myeloma
- CAR-T cel behandeling met Ciltacabtagene autoleucel (cilta-cel-infusie) geeft lager risico op ziekteprogressie of overlijden en meer complete remissies dan standaardzorg bij patiënten met multiple myeloma met lenalidomide resistentie



Plaats een reactie ...
Reageer op "ESO-T01 toegediend in een eenmalige intraveneuze injectie geeft uitstekende resultaten van deze vorm van CAR-T celtherapie bij patienten met Multiple Myeloma (botkanker - Kahler)"