13 maart 2023: Zie ook dit artikel: https://kanker-actueel.nl/ivermectine-een-zogeheten-antiparasitair-middel-wormenkuur-blijkt-wel-degelijk-het-coronavirus-covid-19-zeer-goed-te-kunnen-bestrijden-uit-meta-analyse-van-53-studies-blijkt-zeer-grote-effectiviteit.html
13 maart 2023: Op deze website staan alle serieuzere studies uitgevoerd met ivermectine op volgorde van data gepubliceerd. Wie klikt op een vermeldde studie krijgt de belangrijkste uitkomsten en data van betreffende studie. De laatste update is van 10 maart 2023.
13 maart 2023: Bron: Cureus 15 januari 2023
Een grote prospectieve observatiestudie waarbij de inwoners van Itajaí een Braziliaanse stad met 223.128 inwoners allemaal het gebruik van ivermectine is aangeboden en daarna zijn gevolgd en geanalyseerd op het regelmatig gebruik van ivermectine of geen gebruik werden onderzocht in het of verminderen van besmet raken met het coronavirus - Covid-19. Ivermectine werd aangeboden als een optionele behandeling die gedurende twee opeenvolgende dagen om de 15 dagen moest worden ingenomen in een dosis van 0,2 mg/kg/dag
Uit de studie blijkt dat regelmatig gebruik van ivermectine als profylactisch middel gepaard ging met statistisch significant lagere COVID-19-infecties, ziekenhuisopnames en sterftecijfers en geeft aan dat het nemen van ivermectine mensen beter beschermd tegen het coronavirus dan geen ivermectine nemen.
Van de 223.128 inwoners van Itajaí die in aanmerking kwamen voor de studie, werden in totaal 159.561 proefpersonen opgenomen in de analyse: 113.845 (71,3%) regelmatige ivermectinegebruikers en 45.716 (23,3%) niet-gebruikers. Hiervan waren 4.311 ivermectinegebruikers besmet, waarvan 4.197 uit de stad Itajaí (3,7% infectiepercentage), en 3.034 niet-gebruikers (uit Itajaí) waren besmet (6,6% infectiepercentage), met een vermindering van 44% in COVID -19 infectiepercentage (risicoratio , 0,56; 95% betrouwbaarheidsinterval (95% BI), 0,53-0,58; p < 0,0001).
- Met behulp van de PSM studie werden twee groepen van 3.034 proefpersonen met een COVID-19-infectie vergeleken.
- Het regelmatige gebruik van ivermectine leidde tot een vermindering van 68% in COVID-19-mortaliteit (25 [0,8%] versus 79 [2,6%] onder niet-gebruikers van ivermectine; RR, 0,32; 95% BI, 0,20-0,49; p < 0,0001) . Gecorrigeerd voor restvariabelen was de verlaging van het sterftecijfer 70% (RR, 0,30; 95% BI, 0,19-0,46; p < 0,0001).
- Er was een vermindering van 56% in het aantal ziekenhuisopnames (44 versus 99 ziekenhuisopnames bij respectievelijk ivermectinegebruikers en niet-gebruikers; RR, 0,44; 95% CI, 0,31-0,63; p < 0,0001).
- Na correctie voor resterende variabelen was de vermindering van het aantal ziekenhuisopnames 67% (RR, 0,33; 95% BI, 023-0,66; p < 0,0001).
Conclusies:
In een stadsbreed ivermectineprogramma met profylactisch, optioneel gebruik van ivermectine voor COVID-19, werd ivermectine in verband gebracht met statistisch significant lagere COVID-19-infecties, ziekenhuisopnames en sterftecijfers als gevolg van COVID-19.
Hier een van de vele grafieken uit de studie die aangeeft hoe deze studie was opgezet.
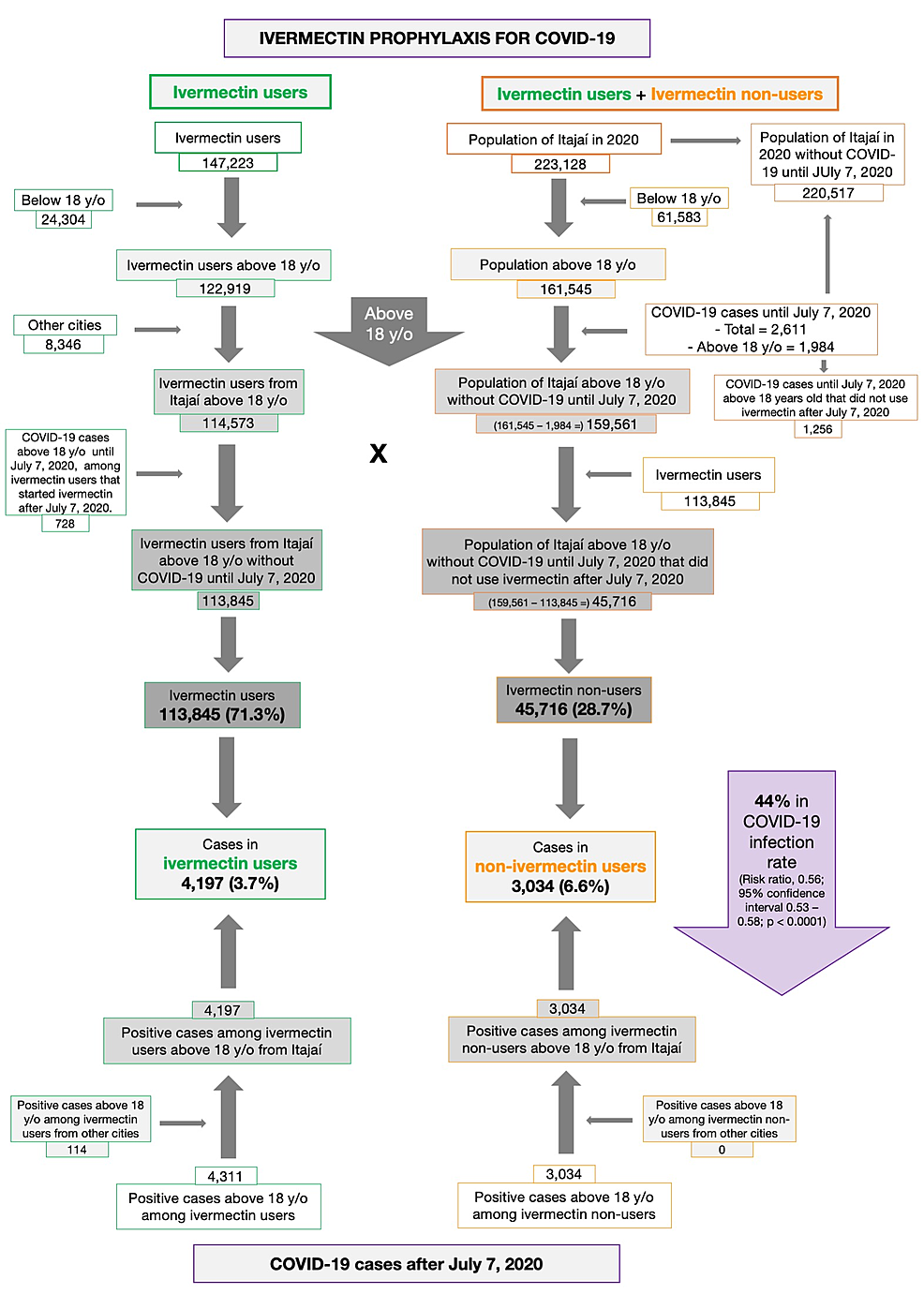
Figure 1: Underlying data for the study on ivermectin prophylaxis used for COVID-19.
Het volledige studieverslag is gratis in te zien. Klik de titel van het abstract:
Published: January 15, 2022
DOI: 10.7759/cureus.21272 
Cite this article as: Kerr L, Cadegiani F A, Baldi F, et al. (January 15, 2022) Ivermectin Prophylaxis Used for COVID-19: A Citywide, Prospective, Observational Study of 223,128 Subjects Using Propensity Score Matching. Cureus 14(1): e21272. doi:10.7759/cureus.21272
Abstract
Background: Ivermectin has demonstrated different mechanisms of action that potentially protect from both coronavirus disease 2019 (COVID-19) infection and COVID-19-related comorbidities. Based on the studies suggesting efficacy in prophylaxis combined with the known safety profile of ivermectin, a citywide prevention program using ivermectin for COVID-19 was implemented in Itajaí, a southern city in Brazil in the state of Santa Catarina. The objective of this study was to evaluate the impact of regular ivermectin use on subsequent COVID-19 infection and mortality rates.
Materials and methods: We analyzed data from a prospective, observational study of the citywide COVID-19 prevention with ivermectin program, which was conducted between July 2020 and December 2020 in Itajaí, Brazil. Study design, institutional review board approval, and analysis of registry data occurred after completion of the program. The program consisted of inviting the entire population of Itajaí to a medical visit to enroll in the program and to compile baseline, personal, demographic, and medical information. In the absence of contraindications, ivermectin was offered as an optional treatment to be taken for two consecutive days every 15 days at a dose of 0.2 mg/kg/day. In cases where a participating citizen of Itajaí became ill with COVID-19, they were recommended not to use ivermectin or any other medication in early outpatient treatment. Clinical outcomes of infection, hospitalization, and death were automatically reported and entered into the registry in real time. Study analysis consisted of comparing ivermectin users with non-users using cohorts of infected patients propensity score-matched by age, sex, and comorbidities. COVID-19 infection and mortality rates were analyzed with and without the use of propensity score matching (PSM).
Results: Of the 223,128 citizens of Itajaí considered for the study, a total of 159,561 subjects were included in the analysis: 113,845 (71.3%) regular ivermectin users and 45,716 (23.3%) non-users. Of these, 4,311 ivermectin users were infected, among which 4,197 were from the city of Itajaí (3.7% infection rate), and 3,034 non-users (from Itajaí) were infected (6.6% infection rate), with a 44% reduction in COVID-19 infection rate (risk ratio , 0.56; 95% confidence interval (95% CI), 0.53-0.58; p < 0.0001). Using PSM, two cohorts of 3,034 subjects suffering from COVID-19 infection were compared. The regular use of ivermectin led to a 68% reduction in COVID-19 mortality (25 [0.8%] versus 79 [2.6%] among ivermectin non-users; RR, 0.32; 95% CI, 0.20-0.49; p < 0.0001). When adjusted for residual variables, reduction in mortality rate was 70% (RR, 0.30; 95% CI, 0.19-0.46; p < 0.0001). There was a 56% reduction in hospitalization rate (44 versus 99 hospitalizations among ivermectin users and non-users, respectively; RR, 0.44; 95% CI, 0.31-0.63; p < 0.0001). After adjustment for residual variables, reduction in hospitalization rate was 67% (RR, 0.33; 95% CI, 023-0.66; p < 0.0001).
Conclusion: In this large PSM study, regular use of ivermectin as a prophylactic agent was associated with significantly reduced COVID-19 infection, hospitalization, and mortality rates.
Final discussion
In this citywide ivermectin prophylaxis program, a large, statistically significant decrease in mortality rate was observed after the program began among the entire population of city residents. When comparing subjects that used ivermectin regularly, non-users were two times more likely to die from COVID-19 while ivermectin users were 7% less likely to be infected with SARS-CoV-2 (p = 0.003).
Although this study is not a randomized, double-blind, placebo-controlled clinical trial, the data were prospectively collected and resulted in a massive study sample that allowed adjustment for numerous confounding factors, thus strengthening the findings of the present study.
Due to the well-established, long-term safety profile of ivermectin, with rare adverse effects, the absence of proven therapeutic options to prevent death caused by COVID-19, and lack of effectiveness of vaccines in real-life all-cause mortality analyses to date, we recommend that ivermectin be considered as a preventive strategy, in particular for those at a higher risk of complications from COVID-19 or at higher risk of contracting the illness, not as a substitute for COVID-19 vaccines, but as an additional tool, particularly during periods of high transmission rates.
Conclusions
In a citywide ivermectin program with prophylactic, optional ivermectin use for COVID-19, ivermectin was associated with significantly reduced COVID-19 infection, hospitalization, and death rates from COVID-19.
References
- Chen IS, Kubo Y: Ivermectin and its target molecules: shared and unique modulation mechanisms of ion channels and receptors by ivermectin. J Physiol. 2018, 596:1833-45. 10.1113/JP275236
- Kaur H, Shekhar N, Sharma S, Sarma P, Prakash A, Medhi B: Ivermectin as a potential drug for treatment of COVID-19: an in-sync review with clinical and computational attributes. Pharmacol Rep. 2021, 73:736-49. 10.1007/s43440-020-00195-y
- Martin RJ, Robertson AP, Choudhary S: Ivermectin: an anthelmintic, an insecticide, and much more. Trends Parasitol. 2021, 37:48-64. 10.1016/j.pt.2020.10.005
- Mastrangelo E, Pezzullo M, De Burghgraeve T, et al.: Ivermectin is a potent inhibitor of flavivirus replication specifically targeting NS3 helicase activity: new prospects for an old drug. J Antimicrob Chemother. 2012, 67:1884-94. 10.1093/jac/dks147
- Wagstaff KM, Sivakumaran H, Heaton SM, Harrich D, Jans DA: Ivermectin is a specific inhibitor of importin α/β-mediated nuclear import able to inhibit replication of HIV-1 and dengue virus. Biochem J. 2012, 443:851-6. 10.1042/BJ20120150
- Crump A: Ivermectin: enigmatic multifaceted 'wonder' drug continues to surprise and exceed expectations. J Antibiot (Tokyo). 2017, 70:495-505. 10.1038/ja.2017.11
- Heidary F, Gharebaghi R: Ivermectin: a systematic review from antiviral effects to COVID-19 complementary regimen. J Antibiot (Tokyo). 2020, 73:593-602. 10.1038/s41429-020-0336-z
- Li N, Zhao L, Zhan X: Quantitative proteomics reveals a broad-spectrum antiviral property of ivermectin, benefiting for COVID-19 treatment. J Cell Physiol. 2021, 236:2959-75. 10.1002/jcp.30055
- Jin L, Feng X, Rong H, et al.: The antiparasitic drug ivermectin is a novel FXR ligand that regulates metabolism. Nat Commun. 2013, 4:1937. 10.1038/ncomms2924
- Yang JS, Qi W, Farias-Pereira R, Choi S, Clark JM, Kim D, Park Y: Permethrin and ivermectin modulate lipid metabolism in steatosis-induced HepG2 hepatocyte. Food Chem Toxicol. 2019, 125:595-604. 10.1016/j.fct.2019.02.005
- Cairns DM, Giordano JE, Conte S, Levin M, Kaplan DL: Ivermectin promotes peripheral nerve regeneration during wound healing. ACS Omega. 2018, 3:12392-402. 10.1021/acsomega.8b01451
- Zheng YY, Ma YT, Zhang JY, Xie X: COVID-19 and the cardiovascular system. Nat Rev Cardiol. 2020, 17:259-60. 10.1038/s41569-020-0360-5
- Nagai H, Satomi T, Abiru A, et al.: Antihypertrophic effects of small molecules that maintain mitochondrial ATP levels under hypoxia. EBioMedicine. 2017, 24:147-58. 10.1016/j.ebiom.2017.09.022
- Park A, Iwasaki A: Type I and type III interferons - induction, signaling, evasion, and application to combat COVID-19. Cell Host Microbe. 2020, 27:870-8. 10.1016/j.chom.2020.05.008
- Zhang X, Song Y, Ci X, et al.: Ivermectin inhibits LPS-induced production of inflammatory cytokines and improves LPS-induced survival in mice. Inflamm Res. 2008, 57:524-9. 10.1007/s00011-008-8007-8
- Okeahialam BN: Serine protease inhibitors could be of benefit in the treatment of COVID-19 disease. Ther Adv Infect Dis. 2021, 8:10.1177/20499361211032048
- Matsuyama T, Kubli SP, Yoshinaga SK, Pfeffer K, Mak TW: An aberrant STAT pathway is central to COVID-19. Cell Death Differ. 2020, 27:3209-25. 10.1038/s41418-020-00633-7
- Wang K, Gao W, Dou Q, Chen H, Li Q, Nice EC, Huang C: Ivermectin induces PAK1-mediated cytostatic autophagy in breast cancer. Autophagy. 2016, 12:2498-9. 10.1080/15548627.2016.1231494
- Dou Q, Chen HN, Wang K, et al.: Ivermectin induces cytostatic autophagy by blocking the PAK1/Akt axis in breast cancer. Cancer Res. 2016, 76:4457-69. 10.1158/0008-5472.CAN-15-2887
- Layhadi JA, Turner J, Crossman D, Fountain SJ: ATP evokes Ca2+ responses and CXCL5 secretion via P2X4 receptor activation in human monocyte-derived macrophages. J Immunol. 2018, 200:1159-68. 10.4049/jimmunol.1700965
- Juarez M, Schcolnik-Cabrera A, Dueñas-Gonzalez A: The multitargeted drug ivermectin: from an antiparasitic agent to a repositioned cancer drug. Am J Cancer Res. 2018, 8:317-31.
- Andersson U, Ottestad W, Tracey KJ: Extracellular HMGB1: a therapeutic target in severe pulmonary inflammation including COVID-19?. Mol Med. 2020, 26:42. 10.1186/s10020-020-00172-4
- Yan S, Ci X, Chen N, et al.: Anti-inflammatory effects of ivermectin in mouse model of allergic asthma. Inflamm Res. 2011, 60:589-96. 10.1007/s00011-011-0307-8
- Reis TA, Oliveira-da-Silva JA, Tavares GS, et al.: Ivermectin presents effective and selective antileishmanial activity in vitro and in vivo against Leishmania infantum and is therapeutic against visceral leishmaniasis. Exp Parasitol. 2021, 221:108059. 10.1016/j.exppara.2020.108059
- Scheim D: Ivermectin for COVID-19 treatment: clinical response at quasi-threshold doses via hypothesized alleviation of CD147-mediated vascular occlusion. . SSRN J. 2020, 10.2139/ssrn.3636557
- Ci X, Li H, Yu Q, et al.: Avermectin exerts anti-inflammatory effect by downregulating the nuclear transcription factor kappa-B and mitogen-activated protein kinase activation pathway. Fundam Clin Pharmacol. 2009, 23:449-55. 10.1111/j.1472-8206.2009.00684.x
- Zaidi AK, Dehgani-Mobaraki P: The mechanisms of action of Ivermectin against SARS-CoV-2: an evidence-based clinical review article. . J Antibiot (Tokyo). 2021, 10.1038/s41429-021-00430-5
- Kalfas S, Visvanathan K, Chan K, Drago J: The therapeutic potential of ivermectin for COVID- 19: a systematic review of mechanisms and evidence. . medRxiv. 2020, 10.1101/2020.11.30.20236570
- Behera P, Patro BK, Singh AK, et al.: Role of ivermectin in the prevention of SARS-CoV-2 infection among healthcare workers in India: a matched case-control study. PLoS One. 2021, 16:e0247163. 10.1371/journal.pone.0247163
- Hellwig MD, Maia A: A COVID-19 prophylaxis? Lower incidence associated with prophylactic administration of ivermectin. Int J Antimicrob Agents. 2021, 57:106248. 10.1016/j.ijantimicag.2020.106248
- World Medical Association. Declaration of Helsinki. (2020). Accessed: December 24, 2021: https://www.wma.net/what-we-do/medical-ethics/declaration-of-helsinki/doh-oct2000/.
- Conselho Federal de Medicina (CFM). Código de Ética Médica. (2010). Accessed: December 24, 2021: https://portal.cfm.org.br/images/stories/biblioteca/codigo%20de%20etica%20medica.pdf.
- Nguyen TL, Collins GS, Spence J, Daurès JP, Devereaux PJ, Landais P, Le Manach Y: Double-adjustment in propensity score matching analysis: choosing a threshold for considering residual imbalance. BMC Med Res Methodol. 2017, 17:78. 10.1186/s12874-017-0338-0
- Zhang Z, Kim HJ, Lonjon G, Zhu Y: Balance diagnostics after propensity score matching. Ann Transl Med. 2019, 7:16. 10.21037/atm.2018.12.10
- Boletim epidemiológico do estado de Santa Catarina, Brasil. (2020). Accessed: December 24, 2021: http://www.dive.sc.gov.br.
- Choudhury A, Das NC, Patra R, Bhattacharya M, Ghosh P, Patra BC, Mukherjee S: Exploring the binding efficacy of ivermectin against the key proteins of SARS-CoV-2 pathogenesis: an in silico approach. Future Virol. 2021, 16:277-91. 10.2217/fvl-2020-0342
- Austin PC: An introduction to propensity score methods for reducing the effects of confounding in observational studies. Multivariate Behav Res. 2011, 46:399-424. 10.1080/00273171.2011.568786
- Propensity score matching in the absence of randomized controlled trials: a case study on the effects of breastfeeding on childhood obesity. (2020). Accessed: December 24, 2021: https://methods.sagepub.com/case/propensity-score-matching-trials-effects-breastfeeding-childhood-obesity.
corona virus, COVID-19, preventie, bescherming, IC opnames, risico op overlijden, ivermectine
Gerelateerde artikelen




Plaats een reactie ...
Reageer op "Ivermectine als profylactisch middel preventief genomen geeft statistisch significant lagere COVID-19-infecties, ziekenhuisopnames en sterftecijfers in vergelijking met geen gebruik van ivermectine"