Mocht u kanker-actueel de moeite waard vinden en ons willen ondersteunen om kanker-actueel online te houden dan kunt u ons machtigen voor een periodieke donatie via donaties: https://kanker-actueel.nl/NL/donaties.html of doneer al of niet anoniem op - rekeningnummer NL79 RABO 0372931138 t.n.v. Stichting Gezondheid Actueel in Amersfoort. Onze IBANcode is NL79 RABO 0372 9311 38
Elk bedrag is welkom. En we zijn een ANBI instelling dus uw donatie of gift is in principe aftrekbaar voor de belasting.
En als donateur kunt u ook korting krijgen bij verschillende bedrijven:
11 december 2015: lees ook dit artikel:
11 december 2015:
Aanvullend op onderstaande informatie hier een gratis in te zien volledig studierapport over de effectiviteit (90% effectieve respons) met goed hanteerbare bijwerkingen bij een chemo embolisatie van borstkanker Resaic geheten: Main complications and results of treatment with intra-arterial infusion chemotherapy through the subclavian and thoracic arteries for locally advanced breast cancer
Abstract met ook interessante referentielijst staat onderaan dit artikel.
Van de inmiddels 53 patienten (allemaal borstkanker stadium IIIa - N = 10 of IIIb N = 24 of IIIc - N = 19) (N = 10, bereikten 7 patiënten een CR - complete remissie (13.2%); 41 patiënten ervaarden een PR - gedeeltelijke remissie (PR = 50% of meer vermindering van tumorgrootte en aantal) (77.4%), met een hazard ratio van effectiviteit samen van (CR + PR: 90.6%, 48/53); 5 patiënten behielden SD = stabiele ziekte (9.4%) en niemand kreeg progressie van de ziekte tijdens de behandelingen.
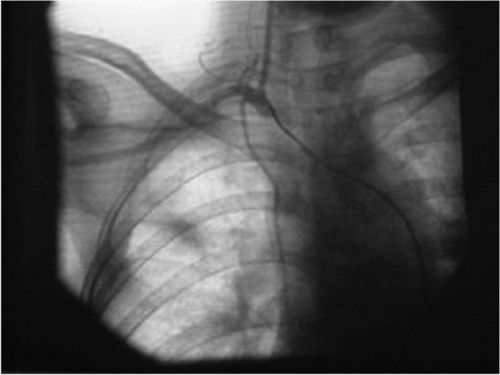
Foto: For patients in stage IIIb and IIIc, the catheter was guided to the opening of the subclavian and lateral thoracic arteries. Prior to drug administration, a contrast agent (iohexol) was injected to comfirm the location of the catheter. An arteriogram ...
19 juli 2011: vorig jaar werden resultaten met Resaic - redistributed subclavian arterial infusion chemotherapy, oftewel chemo embolisatie bij borstkanker naar buiten gebracht. Recent zijn ook de resultaten van een fase I studie bij 22 patienten met chemo resistente uitgezaaide borstkanker gepubliceerd die een significant positief effect te zien geven. Er werd bij 77% alsnog een respons gezien en de toxiciteit was slechts bij 6 van de 22 patienten graad 3 , maar wel ook nog behandelbaar. Op zich dus een bemoedigend resultaat. Wel opvallend dat alleen in Japan in 1 kliniek met deze methode wordt gewerkt. Althans ik kan verder niets vinden in pubmed over deze nieuwe vorm van chemo embolisatie bij gevorderde borstkanker. Onderaan staat nieuwe abstract.
10 januari 2010: Bron: Department of Radiology, St Marianna University School of Medicine, Sugao, Miyamae, Kawasaki, Kanagawa, Japan
Resaic (redistributed subclavian arterial infusion chemotherapy) een vorm van Arteriele chemo embolisatie bij borstkanker geeft bemoedigende resultaten. Dit blijkt uit een fase I/II studie uitgevoerd door Japanse onderzoekers.
Elf patiënten ondergingen RESAIC, en de complete remissie en gedeeltelijke respons op 10 evalueerbare patiënten was 90%: complete remissie n = 4, partiële remissie n = 4, stabiele ziekte, n = 1, en niet evalueerbaar n = 1. Drie van de vier patiënten met CR had geen metastasen op afstand, en de gemodificeerde radicale mastectomie werd uitgevoerd1 maand na het beeindigen van RESAIC.
Het via een biopt genomen weefsel toonde geen resterende kankercellen en bevestigde de complete remissie die eerder werd gediagnosteerd in elk van deze gevallen. Hoewel tijdelijke graad-3 myelosuppressie werd waargenomen bij drie patiënten die eerder zijn behandeld door systemische chemotherapie, waren er geen andere medicijn-gerelateerde bijwerkingen of behandelings gerelateerde complicaties.
RESAIC gaf een betere respons te zien en toonde geen grote complicaties in vergelijking met andere behandelingen, ondanks het vergevorderde stadium van de kanker.
Development of a new subclavian arterial infusion chemotherapy method for locally or recurrent advanced breast cancer using an implanted catheter-port system after redistribution of arterial tumor supply.
Takizawa K, Shimamoto H, Ogawa Y, Yoshimatsu M, Yagihashi K, Nakajima Y, Kitanosono T.
Department of Radiology, St Marianna University School of Medicine, Sugao, Miyamae, Kawasaki, Kanagawa, Japan. taki-lrl@vy.catv.ne.jp
Locally or recurrent advanced breast cancers can receive arterial blood supply from various arteries, such as the internal thoracic artery (ITA), the lateral thoracic artery, and the other small arterial branches originating from the subclavian artery. Failure to catheterize and subsequent formation of collateral arterial blood supply from various arteries are some of the reasons why the response to conventional selective transarterial infusion chemotherapy is limited and variable. To overcome this problem, we developed a new subclavian arterial infusion chemotherapy method using an implanted catheter-port system after redistribution of arterial tumor blood supply by embolizing the ITA. We named this technique ("redistributed subclavian arterial infusion chemotherapy" (RESAIC)). Using RESAIC, patients can be treated on an outpatient basis for extended periods of time. Eleven patients underwent RESAIC, and the complete remission and partial response rate in 10 evaluable patients was 90%: complete remission n = 4, partial remission n = 4, stable disease n = 1, and not evaluable n = 1. Three of four patients with CR had no distant metastasis, and modified radical mastectomy was performed 1 month after conclusion of RESAIC. The resected specimens showed no residual cancer cells, and pathologically confirmed complete remission was diagnosed in each of these cases. Although temporary grade-3 myelosuppression was seen in three patients who were previously treated by systemic chemotherapy, there was no other drug-induced toxicity or procedure-related complications. RESAIC produced a better response and showed no major complications compared with other studies despite the advanced stage of the cancers.
PMID: 19238484 [PubMed - indexed for MEDLINE]
Clinical efficacy and value of redistributed subclavian arterial infusion chemotherapy for locally advanced breast cancer
Bron: Spingerlink
Clinical efficacy and value of redistributed subclavian arterial infusion chemotherapy for locally advanced breast cancer.
Source
Department of Radiology, St. Marianna University School of Medicine, 2-16-1 Sugao, Miyamae-ku, Kawasaki 216-8511, Japan. hshima@auone.jp
Abstract
PURPOSE:
The aim of this study was to evaluate the efficacy and safety of redistributed subclavian arterial infusion chemotherapy (RESAIC). We have focused on the local response, quality of life (QOL), and complications. We have also investigated factors that influence the local response of RESAIC.
MATERIALS AND METHODS:
The subjects were patients with locally advanced breast cancer whose tumors were resistant to standard systemic chemotherapy (at least more than two regimens), those who were physically unable to tolerate systemic chemotherapy, and patients with locally recurrent breast cancer. The registration period was between April 2006 and May 2009.
RESULTS:
A total of 24 cases in 22 patients (mean age 59.5 years, range 36-82 years) were entered in the study. The local response rate of RESAIC was 77.3% (17/22). The QOL score showed improvement on average. There were no serious complications during catheter port implantation, and there was hematological toxicity over grade 3 in 27.3% (6/22) of patients. A significant difference between responders and nonresponders was seen in patients with a replaced type tumor (on imaging, diffuse contrast enhancement was seen in whole quadrants) (P = 0.043), and the patients underwent radiotherapy (P = 0.043).
CONCLUSION:
RESAIC is an effective, safe treatment for locally advanced breast cancer. Because it was reviewed in only a few cases, however, large-scale studies are warranted.
- PMID:
- 21607836
- [PubMed - in process]
intra-arterial infusion chemotherapy is an effective local control treatment for LABC - locally advanced breast cancer
Main complications and results of treatment with intra-arterial infusion chemotherapy through the subclavian and thoracic arteries for locally advanced breast cancer.
Abstract
Intra-arterial infusion chemotherapy for locally advanced breast cancer (LABC) has been previously performed. However, the main complications of this type of chemotherapy remain to be clarified. In the present study, catheterization chemotherapy was carried out for 53 LABC cases (stage IIIa-IIIc) between May, 2006 and March, 2007. For IIIB and IIIC patients, the catheters were guided to the opening of the subclavian artery. For stage IIIa patients, the catheters were placed into the thoracic artery through a subcutaneous femoral artery puncture. One to four cycles of chemotherapy (mean, 1.6 cycles) were administered for the patients using taxotere, epidoxorubicin, 5-fluorouracil and/or cyclophosphamide. The interval time between the two cycles was 21 days. Seven cases were identified as complete response (CR, 13.2%), 41 cases were partial response (PR, 77.4%) with a rate of effectiveness of (CR + PR, 90.6%), 5 cases were stable disease (SD, 9.40%) and no case was progressive. Pain of the ipsilateral upper extremity was present in 7 cases. Two cases exhibited ipsilateral upper extremity atrophy following drug administration from the opening of the subclavian artery. One case experienced neck pain and headache, while in one case necrosis of local skin was evident. Hematological toxicity over grade 3 was observed in 6 cases (11.30%). Systemic toxicity was mild and did not affect the quality of life of the patients. Overall survival was identified as 18/51 (35.3%), and free-disease survival as 10/51 (19.6%). In conclusion, intra-arterial infusion chemotherapy is an effective local control treatment for LABC. The main complications are pain of the ipsilateral upper extremity and neck as well as headache. Severe complications are ipsilateral upper extremity atrophy and necrosis of local skin. During the treatment, controlling the pressure of the tourniquet and velocity of drug administration are crucial for reducing local complications.
KEYWORDS:
efficacy and complication; intra-arterial chemotherapy; locally advanced breast cancer; subclavian artery; thoracic artery
- PMID:
- 24649239
- PMCID:
- PMC3915647
-
References
1. Singletary SE, Allred C, Ashley P, et al. Revision of the American Joint Committee on Cancer staging system for breast cancer. J Clin Oncol. 2002;20:3628–3636. [PubMed]2. Kaufmann M, von Minckwitz G, Bear HD, et al. Recommendations from an international expert panel on the use of neoadjuvant (primary) systemic treatment of operable breast cancer: new perspectives 2006. Ann Oncol. 2007;18:1927–1934. [PubMed]3. Olson JE, Neuberg D, Pandya KJ, et al. The role of radiotherapy in the management of operable locally advanced breast carcinoma: results of a randomized trial by the Eastern Cooperative Oncology Group. Cancer. 1997;79:1138–1149. [PubMed]4. Murakami M, Kuroda Y, Nishimura S, et al. Intraarterial infusion chemotherapy and radiotherapy with or without surgery for patients with locally advanced or recurrent breast cancer. Am J Clin Oncol. 2001;24:185–191. [PubMed]5. Kitagawa K, Yamakado K, Nakatsuka A, et al. Preoperative transcatheter arterial infusion chemotherapy for locally advanced breast cancer (stageIIIb) for down-staging and increase of respectability. Eur J Radiol. 2002;43:31–36. [PubMed]6. Fiorentini G, Tsetis D, Bernardeschi P, et al. First-line intra-arterial chemotherapy (IAC) with epirubicin and mitoxantrone in locally advanced breast cancer. Anticancer Res. 2003;23:4339–4345. [PubMed]7. Pacetti P, Mambrini A, Paolucci R, et al. Intra-arterial chemotherapy: a safe treatment for elderly patients with locally advanced breast cancer. In Vivo. 2006;20:761–764. [PubMed]8. Shimamoto H, Takizawa K, Ogawa Y, et al. Clinical efficacy and value of redistributed subclavian arterial infusion chemotherapy for locally advanced breast cancer. Jpn J Radiol. 2011;29:236–243. [PubMed]9. Shenkier T, Weir L, Levine M, et al. Steering committee on clinical practice guidelines for the care and treatment of breast cancer. Clinical practice guidelines for the care and treatment of breast cancer: 15. Treatment for women with stage III or locally advanced breast cancer. CMAJ. 2004;170:983–994. [PMC free article] [PubMed]10. Huang EH, Tucker SL, Strom EA, et al. Predictors of locoregional recurrence in patients with locally advanced breast cancer treated with neoadjuvant chemotherapy, mastectomy, and radiotherapy. Int J Radiat Oncol Biol Phys. 2005;62:351–357. [PubMed]11. Fisher B, Bryant J, Wolmark N, et al. Effect of preoperative chemotherapy on the outcome of women with operable breast cancer. J Clin Oncol. 1998;16:2672–2685. [PubMed]12. Wolmark N, Wang J, Mamounas E, et al. Preoperative chemotherapy in patients with operable breast cancer: nine-year results from National surgical adjuvant breast and bowel project B-18. J Natl Cancer Inst Monogr. 2001;30:96–102. [PubMed]13. van der Hage JA, van de Velde CJ, Julien JP, et al. Preoperative chemotherapy in primary operable breast cancer: results from the European organization for Research and Treatment of Cancer trial 10902. J Clin Oncol. 2001;19:4224–4237. [PubMed]14. Mauriac L, MacGrogan G, Avril A, et al. Neoadjuvant chemotherapy for operable breast carcinoma larger than 3 cm: a unicentre randomized trial with a 124-month medial follow-up. Institut Bergonié Bordeaux Groupe Sein (IBBGS) Ann Oncol. 1999;10:47–52. [PubMed]15. Eniu A, Palmieri FM, Perez EA. Weekly administration of docetaxel and paclitaxel in metastatic or advanced breast cancer. Oncologist. 2005;10:665–685. [PubMed]16. Heller W, Mazhar D, Ward R, et al. Neoadjuvant 5-fluorouracil epirubicin and cyclophosphamide chemotherapy followed by docetaxel in refractory patients with locally advanced breast cancer. Oncol Rep. 2007;17:253–259. [PubMed]
Articles from Molecular and Clinical Oncology are provided here courtesy of Spandidos Publications
Gerelateerde artikelen
- Chemotherapie na operatie bij oudere vrouwen (70+) met niet uitgezaaide triple negatieve borstkanker hebben betere overlevingsresultaten in vergelijking met de patienten die geen chemotherapie hadden gehad.
- Xeloda (capecitabine) en fluorouracil (5-FU) kunnen levensbedreigende bijwerkingen veroorzaken bij kankerpatienten met een DPD-deficiëntie. FDA pleit voor testen op genmutaties vooraf aan behandelingen. copy 1
- Capecitabine - Xeloda geeft betere mediane overleving dan chemo op taxanen basis bij hormoonresistente uitgezaaide borstkanker type invasief lobulair carcinoom
- Archief chemo en borstkanker artikelen
- Mediane overleving van oudere vrouwen (65 plus) die chemo weigeren voor hun borstkanker (ER pos. HER2 neg.) is minder in vergelijking met vrouwen die wel chemo nemen (3 tot 7 procent op 5-jaars meting).
- Hoge dosis chemotherapie plus (bloed) stamceltransplantatie versus standaard dosis chemotherapie geeft betere overall overleving (+33 procent) maar alleen voor patienten met stadium III borstkanker met meer dan 10 uitzaaiingen in de lymfklieren
- moleculair jodium voor en naast chemotherapie voor borstkanker stadium II en III geeft 36 procent (46 vs 82 procent) betere 5 jaars ziektevrije overleving. En beduidend minder bijwerkingen. copy 1
- S-1 (combinatie van tegafur, gimeracil, en oteracil) geeft betere resultaten dan chemo met taxanen bij eerstelijns behandeling van uitgezaaide borstkanker ( HER2-pos.)
- Chemo bij beginnende operabele hormoongevoelige borstkanker (ER en PR pos en HER2 neg) blijkt bij 70 procent zinloos te zijn. Bij slechts 30 procent heeft chemo enig effect. Blijkt uit grote studie van Oncogen-DX 21 genen test.
- Hormoontherapie vooraf aan operatie geeft dezelfde resultaten op overleving dan chemo vooraf aan operatie maar met minder bijwerkingen bij ER pos. borstkanker.
- Chemo voor borstkanker met type Luminal A (hormoongevoelig met ER en PR pos. en Her2 neg) is zinloos en geeft geen enkel verschil in 10-jaars overleving in vergelijking met geen chemo biij vrouwen in leeftijd voor de overgang copy 1
- Adjuvante chemotherapie gegeven om de 2 weken verbetert totale overleving op 10 jaar met mediaan 29 procent in vergelijking met chemotherapie gegeven op een standaard schema van 3 weken.
- Xeloda - capecitabine aanvullend op chemo of hormoontherapie postoperatief voor borstkanker HER2 neg. na pre operatieve chemo geeft 31 procent minder kans op recidief
- Anti-HER2 medicijnen zoals trastuzumab - herceptin, lapatinib en pertuzumab naast chemo geven beste resultaten op complete remissies en ziektevrije tijd
- Resaic, een vorm van arteriele chemo embolisatie bij borstkanker geeft uitstekende resultaten zonder bijwerkingen. Fase I studie met 22 patienten bevestigt goede resultaten
- Pertuzumab toegevoegd aan herceptin - trastuzumab en docetaxel verbetert significant ziektevrije tijd en overall overleving, aldus Cleopatra studie
- Eribuline - Mesylate door FDA goedgekeurd als medicijn bij uitgezaaide borstkanker geeft 2 1/2 maand extra mediane overlevingstijd
- 4 x chemo bij niet erfelijke operabele borstkanker geeft zelfde overall overleving 95 procent - en ziektevrije tijd 90 procent dan 6 x chemo
- Chemo: Xeloda - capecitabine geeft mediaan significant langere overlevingstijd met minder bijwerkingen bij gevorderde borstkanker dan CMF - cyclophosphamide, methotrexate en fluorouracil
- Chemo: Vinorelbine lijkt de beste chemo naast Herceptin voor borstkanker met Her2-Neu positieve expressie. Vinorelbine geeft minder ernstige bijwerkingen t.o.v. taxotere
- Chemokuren met Adriamycin (doxorubin) en andere zogeheten anthracycline chemo's (epirubicine en idarubicin) voor borstkankerpatienten geeft bij beperkt aantal (ca. 20%) een positief effect. NCI raadt chemo gevoleigheidstest aan
- Chemo bij borstkanker: Doxorubicin verpakt in liposomen - vetbolletjes geeft iets beter effect maar met beduidend minder bijwerkingen dan intraveneus chemo bij borstkankerpatienten.
- TACE - Transarteriële chemo-embolisatie - bij borstkankerpatiënten voor hun leveruitzaaiïngen verlengt het leven van deze patiënten significant.
- Paclitaxel verpakt in speciaal verpakte vetbolletjes (liposomen - Nab-paclitaxel) geeft significant langere ziektevrije tijd en minder bijwerkingen dan taxol intraveneus bij borstkanker.
- Chemo bij borstkanker: hier een overzicht van belangrijke artikelen en recente studies bij elkaar gezet.



Plaats een reactie ...
Reageer op "Resaic, een vorm van arteriele chemo embolisatie bij borstkanker geeft uitstekende resultaten zonder bijwerkingen. Fase I studie met 22 patienten bevestigt goede resultaten"